Polysaccharide chemistry is enjoying accelerating development thanks to advances in synthetic techniques, biochemistry and solvents, which enable polysaccharide materials to be useful in a variety of demanding applications. Among the synthetic advances, click chemistry has reconfigured the realm of polysaccharide modification that previously was dominated by conventional synthetic approaches such as esterification and etherification. “Click” reactions provide mild, modular, and efficient modification pathways, and equally importantly allow us to synthesize derivatives with novel functionality, architecture, and properties, that are otherwise difficult to obtain via conventional methods. Herein, we review application in polysaccharide modification of six groups of click reactions; CuAAC (copper catalyzed alkyne/azide cycloaddition), metal-free [3+2] cycloaddition, Diels–Alder reaction, oxime click, thiol-Michael reaction, and thiol-ene reaction, as well as one click-like reaction that is the subject of our own research, olefin cross-metathesis.
Copyright © 2015 Elsevier Ltd. All rights reserved.
 |
click reaction review |
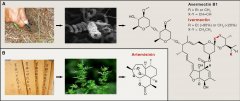
Ben Shen DOI: http://dx.doi.org/10.1016/j.cell.2015.11.031 The 2015 Nobel Prize in Physiology or Medicine has been awarded to William C. Campbell, Satoshi Omura, and Youyou Tu for t...

Xiangtao Meng a , b , Kevin J. Edgar a , b , , a Macromolecules and Interfaces Institute, Virginia Tech, Blacksburg, VA 24061, United States b Department of Sustainable Biomaterials...

遗传, 2018, 40(10): 874-887 doi: 10.16288/j.yczz.18-169 综述 中国丝状真菌次级代谢分子调控研究进展 潘园园,1, 刘钢,1,2 1. 中国科学院微生物研究所,真...

Self-Fueled Biomimetic Liquid Metal Mollusk Authors Jie Zhang, Youyou Yao, Lei Sheng, Jing Liu Abstract A liquid metal motor that can eat aluminum food and then move spontaneously a...